Leverage advanced modeling and simulation to reduce costs, accelerate the regulatory approval process, and improve patient outcomes.
Map Unavailable
Date/Time
Date(s) - May 12, 2022
1:00 pm - 2:00 pm
Categories

Medical device development costs continue to rise due to increasing product complexity and tougher FDA regulations. However, in recent years, regulatory agencies have begun to recognize simulation-based product development and virtual prototyping as valid tools to supplement clinical trials, and leading health care companies are adopting these tools to optimize device performance and reliability while simultaneously reducing costs and time to market.
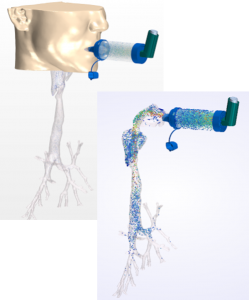
This webinar will discuss how Siemens’ multiphysics simulation solutions enable product developers to generate digital evidence of device performance and improve safety and reliability. Join us to discover how computational fluid dynamics (CFD) with Simcenter STAR-CCM+ and system simulation with Simcenter Amesim can be leveraged in parallel with experimental testing to develop superior products, reduce costs, and accelerate regulatory approval.
Presenters
– Adam Green, ATA Engineering, Inc.
– Mark Carlson, Siemens Digital Industries Software

